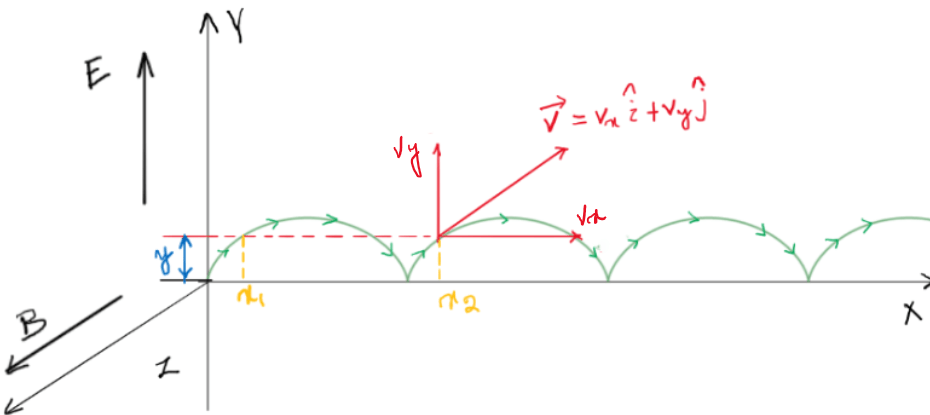
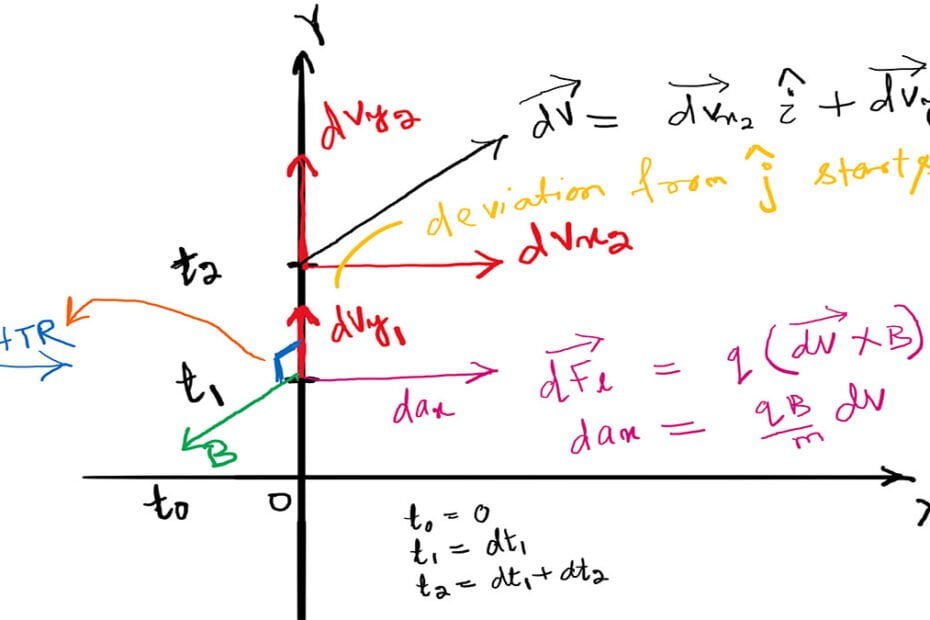
Which is the most stable nuclide?
Which is the most stable nuclide? The popular verdict says $^{56} _{26}Fe$ is the most stable nuclide since it has the highest binding energy per nucleon $(\frac{B}{A}=8.790 \ MeV/A)$. However, this is incorrect. The most stable of all the nuclides is $^{62} _{28}Ni$. Its binding energy per nucleon is $(\frac{B}{A}=8.794… Read More »Which is the most stable nuclide?
Visit Post